View a brief video to better understand how mild® works and what to expect during and after the procedure.
View Educational Video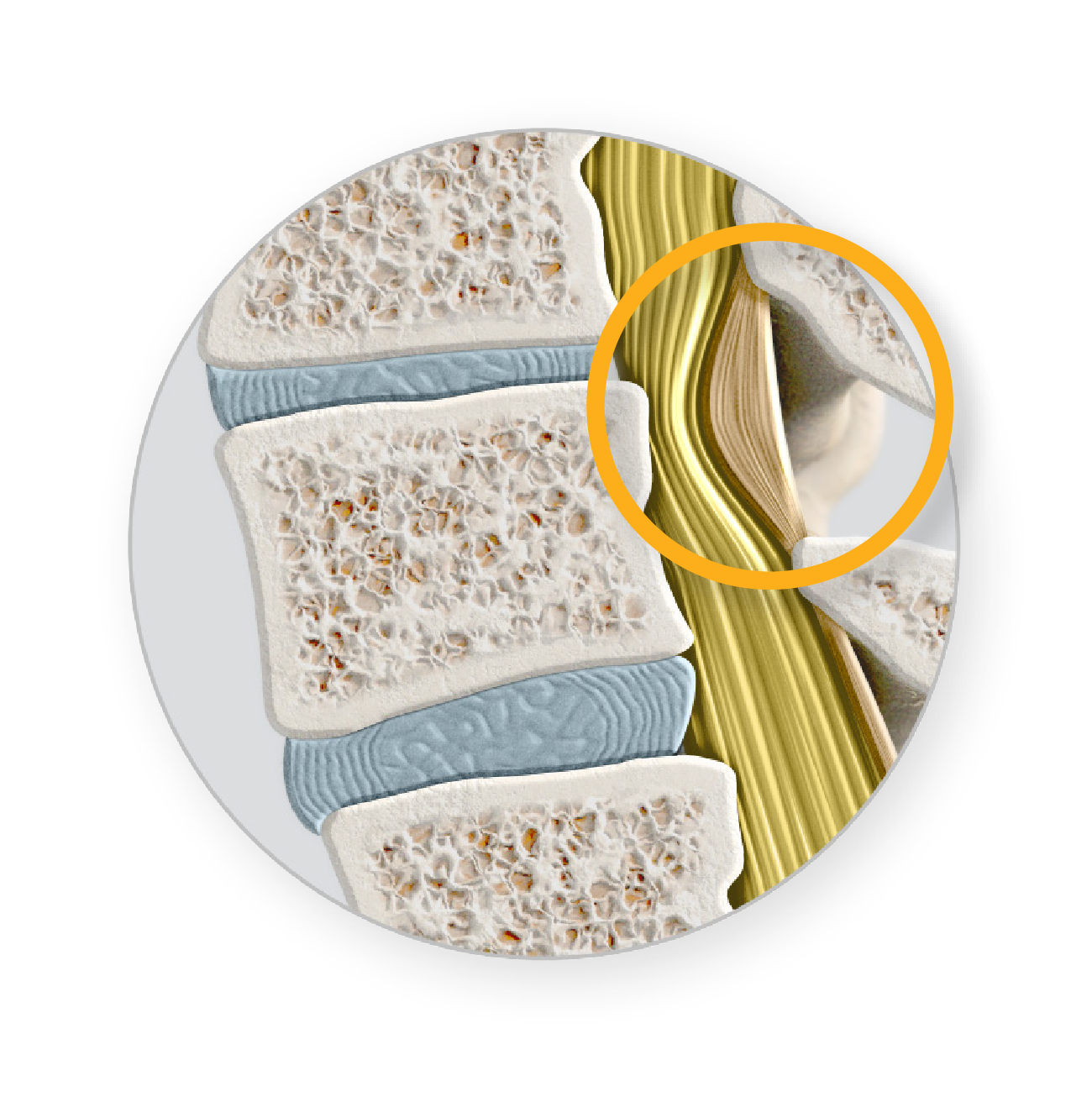

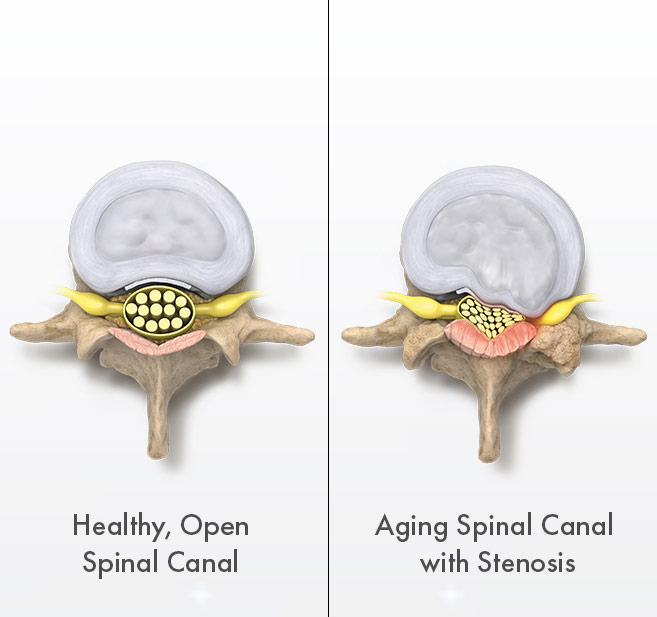
The natural wear and tear on the spine as people age can lead to a number of contributing factors that cause the narrowing of the spinal canal and create pressure on the spinal nerves:

In addition to taking a medical history that includes a list of your symptoms, other tests may be performed to verify LSS:



mild® is an early treatment option to consider when conservative therapies (e.g., physical therapy, pain medication, chiropractic) are not providing adequate relief. The mild® Procedure addresses a major root cause of LSS by removing excess ligament tissue to restore space in the spinal canal. The mild® Procedure typically takes less than an hour and can be performed through a single, tiny incision smaller than the size of a baby aspirin (5.1 mm).
Whether you were recently diagnosed with LSS, have had surgery or are getting epidural steroid injections that are no longer providing pain relief, talk to a mild® Doctor to see if you are a candidate for the mild® Procedure.

Cleveland Clinic 1-Year Study: Patients were able to increase their average standing time from 8 minutes to 56 minutes with less pain.

Cleveland Clinic 1-Year Study: Patients were able to increase their average walking distance from 246 feet to 3,956 feet with less pain.

Cleveland Clinic 5-Year Study: mild® helped 88% of patients avoid back surgery for at least 5 years while providing lasting relief.
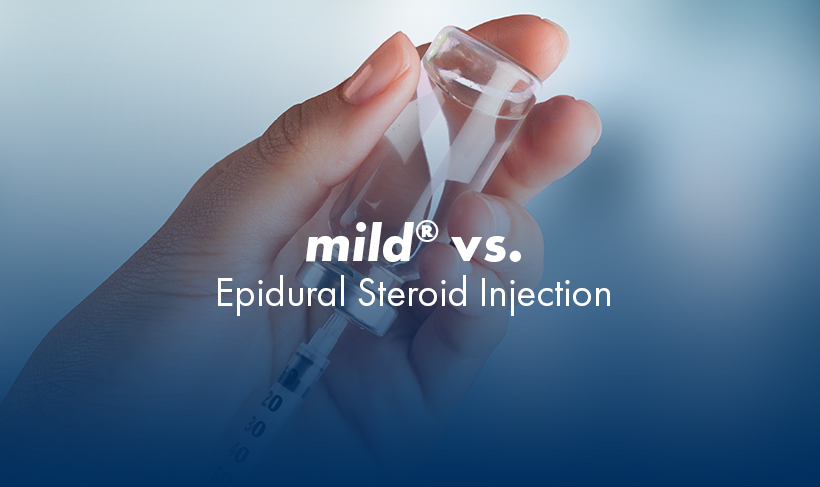
MiDAS ENCORE 2-Year Study: mild® has a safety profile similar to an epidural steroid injection, but with lasting results. In this study, there were no statistically significant differences in the safety profile between study groups.
mild® is a minimally invasive procedure typically performed in an outpatient setting using only local anesthetic and light sedation. The nature of the procedure carries that do not apply to receiving an epidural steroid injection.

MiDAS ENCORE 2-Year Study: Patients reported an 85% patient satisfaction rate with the mild® Procedure.
The mild® Procedure is designed to get you in and out and on your way. mild® does not require general anesthesia, implants, stitches, steroids, or opioids. The procedure can be performed using local anesthetic and light sedation and leaves no implants behind, only a Band-Aid.
Patients typically resume normal activity within 24 hours with no restrictions
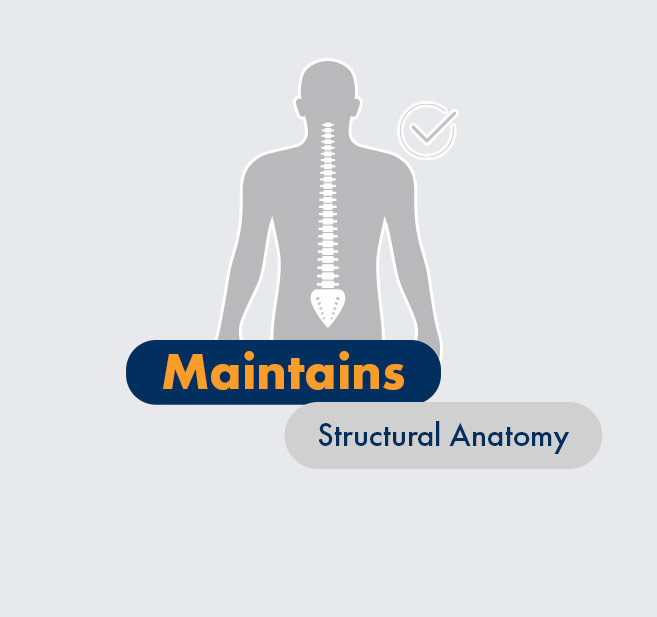
mild® addresses a major root cause of LSS by removing small pieces of bone and thickened ligament. This restores space in the spinal canal, which reduces the compression of the nerves.
Unlike some major surgeries, mild® doesn’t eliminate future treatment options—because with mild®, no major structural anatomy of the spine is altered.
The mild® Procedure is covered nationwide by Medicare and Medicare Advantage. Commercial coverage and other plans vary. Talk to your doctor to obtain coverage specifics for your plan type.
Complete a short survey to determine if mild® may be an effective treatment option for you. Print or save your results and discuss with a mild® Doctor.
Take the mild® SurveyPain or numbness in lower back when standing
Pain, numbness or tingling in legs or buttocks when walking
Relief when sitting or leaning forward



Are you one of over two million patients suffering from lumbar spinal stenosis (LSS) annually nationwide? If so, you’re likely experiencing pain or numbness in your lower back when standing upright; pain, numbness, heaviness or tingling in your upper legs or buttocks when walking and pain when sleeping. The pain can become more common the older you get. In fact, lumbar spinal stenosis (LSS) is prevalent in about 20% of patients ages 60+ nationwide. Temporary relief is often found when sitting or bending forward, or by curling into a fetal position when lying down.
There are, however, other ways to relieve this pain and get back to what matters to you most! If your current treatment for lumbar spinal stenosis (LSS) isn’t providing you adequate relief, consider minimally invasive lumbar decompression (mild®) as your next step.
Benyamin RM, Staats PS, MiDAS ENCORE Investigators. mild® is an effective treatment for lumbar spinal stenosis with neurogenic claudication: MiDAS ENCORE Randomized Controlled Trial. Pain Physician. 2016;19(4):229-242.
Mekhail N, Costandi S, Abraham B, Samuel SW. Functional and patient-reported outcomes in symptomatic lumbar spinal stenosis following percutaneous decompression. Pain Pract. 2012;12(6):417-425. doi:10.1111/j.1533-2500.2012.00565.x.
2012 data from Health Market Sciences report for Vertos Medical 2013.
Data on file with Vertos Medical.
Staats PS, Chafin TB, Golvac S, et al. Long-term safety and efficacy of minimally invasive lumbar decompression procedure for the treatment of lumbar spinal stenosis with neurogenic claudication: 2-year results of MiDAS ENCORE. Reg Anesth Pain Med. 2018;43:789-794. doi:10.1097/AAP.0000000000000868.
Based on mild® Procedure data collected in all clinical studies. Major complications are defined as dural tear and blood loss requiring transfusion.
MiDAS ENCORE responder data. On file with Vertos Medical.
Jain S, Deer TR, Sayed D, et al. Minimally invasive lumbar decompression: a review of indications, techniques, efficacy and safety. Pain Manag. 2020;10(5). https://doi.org/10.2217/pmt-2020-0037. Accessed June 1, 2020.
Deer TR, Grider JS, Pope JE, et al. The MIST Guidelines: the Lumbar Spinal Stenosis Consensus Group guidelines for minimally invasive spine treatment. Pain Pract. 2019;19(3)250-274. doi:10.1111/papr.12744.
Hansson T, Suzuki N, Hebelka H, Gaulitz A. The narrowing of the lumbar spinal canal during loaded MRI: the effects of the disc and ligamentum flavum. Eur Spine J. 2009;18(5):679-686. doi:10.1007/s00586-009-0919-7.
Treatment options shown are commonly offered once conservative therapies (e.g., physical therapy, pain medications, chiropractic) are not providing adequate relief. This is not intended to be a complete list of all treatments available. Doctors typically recommend treatments based on their safety profile, typically prioritizing low risk/less aggressive procedures before higher risk/more aggressive procedures, but will determine which treatments are appropriate for their patients.
The mild® Procedure is a minimally invasive treatment for lumbar spinal stenosis. As with most surgical procedures, serious adverse events, some of which can be fatal, can occur, including heart attack, cardiac arrest (heart stops beating), stroke, and embolism (blood or fat that migrates to the lungs or heart). Other risks include infection and bleeding, spinal cord and nerve injury that can, in rare instances, cause paralysis. This procedure is not for everyone. Physicians should discuss potential risks with patients. For complete information regarding indications for use, warnings, precautions, and methods of use, please reference the devices’ Instructions for Use.
Patient stories on this website reflect the results experienced by individuals who have undergone the mild® Procedure. Patients are not compensated for their testimonial. The mild® Procedure is intended to treat lumbar spinal stenosis (LSS) caused by ligamentum flavum hypertrophy. Although patients may experience relief from the procedure, individual results may vary. Individuals may have symptoms persist or evolve or other conditions that require ongoing medication or additional treatments. Please consult with your doctor to determine if this procedure is right for you.
Reimbursement, especially coding, is dynamic and changes every year. Laws and regulations involving reimbursement are also complex and change frequently. Providers are responsible for determining medical necessity and reporting the codes that accurately describe the work that is done and the products and procedures that are furnished to patients. For this reason, Vertos Medical strongly recommends that you consult with your payers, your specialty society, or the AMA CPT regarding coding, coverage and payment.
Vertos Medical cannot guarantee coding, coverage, or payment for products or procedures. View our Billing Guide.
Vertos is an equal employment opportunity workplace committed to pursuing and hiring a diverse workforce. We strive to grow our team with highly skilled people who share our culture and values. All qualified applicants will receive consideration for employment without regard to sex, age, color, race, religion, marital status, national origin, ancestry, sexual orientation, gender identity, physical & mental disability, medical condition, genetic information, veteran status, or any other basis protected by federal, state or local law.
Hall S, Bartleson JD, Onofrio BM, Baker HL Jr, Okazaki H, O’Duffy JD. Lumbar spinal stenosis. Clinical features, diagnostic procedures, and results of surgical treatment in 68 patients. Ann Intern Med. 1985;103(2):271-275. doi:10.7326/0003-4819-103-2-271.
Kalichman L, Cole R, Kim DH, et al. Spinal stenosis prevalence & association with symptoms: The Framingham Study. Spine J. 2009;9(7):545-550. doi:10.1016/j.spinee.2009.03.005.
Fukusaki M, Kobayashi I, Hara T, Sumikawa K. Symptoms of spinal stenosis do not improve after epidural steroid injection. Clin J Pain. 1998;14(2):148-151. doi:10.1097/00002508-199806000-00010.
Mekhail N, Costandi S, Nageeb G, Ekladios C, Saied O. The durability of minimally invasive lumbar decompression procedure in patients with symptomatic lumbar spinal stenosis: Long-term follow-up [published online ahead of print, 2021 May 4]. Pain Pract. 2021;10.1111/papr.13020. doi:10.1111/papr.13020
Friedly JL, Comstock BA, Turner JA, et al. Long-Term Effects of Repeated Injections of Local Anesthetic With or Without Corticosteroid for Lumbar Spinal Stenosis: A Randomized Trial. Arch Phys Med Rehabil. 2017;98(8):1499-1507.e2. doi:10.1016/j.apmr.2017.02.029
Pope J, Deer TR, Falowski SM. A retrospective, single-center, quantitative analysis of adverse events in patients undergoing spinal stenosis with neurogenic claudication using a novel percutaneous direct lumbar decompression strategy. J Pain Res. 2021;14:1909-1913. doi: 10.2147/JPR.S304997
Pryzbylkowski P, Bux A, Chandwani K, et al. Minimally invasive direct decompression for lumbar spinal stenosis: impact of multiple prior epidural steroid injections [published online ahead of print, 2021 Aug 4]. Pain Manag. 2021;10.2217/pmt-2021-0056. doi:10.2217/pmt-2021-0056
Abstract presented at: American Society of Pain and Neuroscience Annual Conference; July 22-25, 2021; Miami Beach, FL.
Mobility Matters: Low Back Pain in America, Harris Poll Survey, 2022. View data and full summary here.
Deer TR, Grider JS, Pope JE, et al. Best Practices for Minimally Invasive Lumbar Spinal Stenosis Treatment 2.0 (MIST): Consensus Guidance from the American Society of Pain and Neuroscience (ASPN). J Pain Res. 2022;15:1325-1354. Published 2022 May 5. doi:10.2147/JPR.S355285.