Three Signs You May Be Ready to Move to the mild® Procedure
Published June 21, 2021
If you’ve received an epidural steroid injection (ESI) to help manage the pain associated with lumbar spinal stenosis (LSS), but did not experience significant or lasting pain reduction, you are not alone. Although ESIs can be an effective early treatment option for patients with LSS, they are not a one-size-fits-all solution. After an initial epidural injection (or even a series of several injections), many people do not feel significant pain reduction, or many only feel better for a few weeks before the pain returns.
Don’t lose hope—you still have options
Epidurals may not have worked for you, but that does not mean surgery is your only option. Other minimally invasive treatment alternatives, like the mild® Procedure, offer a safe and effective approach that can get you on the path to relief.
mild® has helped thousands of LSS patients achieve lasting relief from back pain.
The mild® Difference: Addressing a Major Root Cause of Your LSS Pain
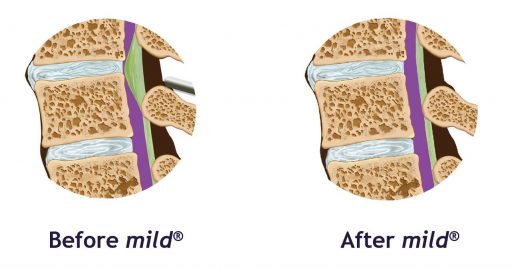
When you have spinal stenosis, your lower spinal canal narrows and compresses the spinal nerves in your lower back. This compression can contribute to the pain and mobility issues you are likely experiencing.
In people with LSS, up to 85% of spinal canal narrowing may be caused by a ligament in the back that becomes thickened over time. The mild® Procedure is a minimally invasive treatment option that removes excess ligament tissue to restore space in the spinal canal. The mild® Procedure typically takes less than an hour, and can be performed through a single, tiny incision smaller than the size of a baby aspirin (5.1 mm).
Are You Ready to Move to mild®?
Patients who have been recommended for, or have tried epidural steroid injections may wonder when they should seriously consider an alternative treatment option that can offer effective, lasting pain relief for their LSS. Here are the 3 most common signs that you may be ready to move to mild®, and the steps you can take if you would like to discuss your options with a mild®-trained doctor.
#1: Your first ESI did not significantly reduce your pain
The patients with LSS who are going to benefit from an ESI can typically be identified fairly quickly after the first injection. In fact, a large clinical study completed across several treatment centers found that the level of pain relief patients experienced 6 weeks after their first injection were generally maintained through 12 months, and that repeated injections offered no additional long-term benefit. Stated simply, 6 weeks after your first injection, you can generally tell whether ESIs are likely to work for you.
Jane Hartigan, an Advanced Practice Provider who works as a Physician Assistant (PA) at a leading pain management practice in Northern California, confirms that their practice routinely moves patients to the mild® Procedure if the first epidural fails. “Our patients love mild®, and it’s been a game-changer in our practice,” Jane says. “mild® offers a similar safety profile to ESIs but with long-term results. So we’ll start with an epidural injection, but we’ll schedule a follow-up soon after the injection to determine if it’s working for that patient. In the many cases that patients are not getting pain relief or improvement in their mobility, we will start to seriously consider the mild® Procedure.”
“mild® offers a similar safety profile to ESIs but with long-term results” -Jane Hartigan, Advanced Practice Provider
#2: You’re feeling “epidural exhaustion” after a series of ESIs
Many patients who receive a series of epidural injections can develop “Epidural Exhaustion,” especially if the injections provide little relief or decreasing relief over time. Jane says, “Epidural Exhaustion is incredibly common in lumbar spinal stenosis patients. A lot of patients come to us from other practices where they received injection after injection with very little improvement. They’re frustrated and can begin to lose hope that they’ll ever feel better.”
In Jane’s practice, LSS patients can move to mild® within weeks of their first injection if the epidural does not relieve their pain. According to Jane, “Many practices perform multiple ESIs, one after another, but we move more quickly toward mild®. When patients are given multiple injections, I feel like it’s just delaying a much better treatment option for that patient. mild® addresses a major root cause of LSS, it’s proven safe and effective, and our patients have had such amazing results. Seeing patients come in after their mild® Procedure, and they’re just so grateful and excited about finally feeling better and being able to walk or stand. It’s so rewarding for me, and reinforces that we’re taking the right approach.”
#3: You’re looking for long-term relief
A key benefit of the mild® Procedure is that unlike ESIs, mild® offers patients lasting results. Many stenosis patients who do show improvement after an initial epidural injection will notice that the relief is short-lived, and additional injections are needed to sustain relief. This is because ESIs do not address the major root cause of LSS. mild®, on the other hand, removes excess ligament tissue that causes the narrowing of the spinal canal without significantly changing the structural anatomy of the spine.
A recent study conducted by the Cleveland Clinic highlights the long-term relief provided by mild®.
The study found:

“One of the most rewarding aspects of offering mild® in our practice is having patients that come back to us with other issues, but their lower back is still feeling better,” noted Jane. “I’ve had patients come in months and years later, and they feel the same relief they experienced after the mild® Procedure. They have gone on with their lives without worrying about their LSS.”
Making the Move to mild®
If you are ready to move on from epidural steroid injections, and to find out if you are a candidate for the mild® Procedure, there are a few steps you can take to put yourself on the path to relief.
Find a mild® doctor in your area
- mild® doctors are specialists who have received specific training on this new, advanced therapy. To search for a doctor in your area, visit our online “Find a mild® Doctor” tool.

Take charge of your health
- Before your next visit with your pain specialist, complete this short survey to determine if minimally invasive lumbar decompression may be an effective treatment option for you.
- Print or save your results and discuss them during your next visit with your doctor or their staff. Don’t be afraid to ask questions and be direct about your goals for your LSS treatment.

Understand the plan
- If you are currently considering or scheduled for another ESI, talk to your pain specialist about considering mild® as your next step. Ask for a complete treatment plan so you understand what will happen if your next ESI fails, or produces only short-term relief.


