In December 2023, Jim from Mentor, OH, was suffering pain related to lumbar spinal stenosis (LSS). At the time, Jim’s mobility was limited. “Standing was minimal,” Jim recalls. “I could hardly walk.”
To address Jim’s pain and mobility challenges, Dr. Hassan Aboumerhi with UH Hospitals recommended the mild® Procedure.
According to Jim, Dr. Aboumerhi described the procedure as simple and similar to receiving an epidural steroid injection. Jim confirms that the procedure was short. “The mild® Procedure was perfect for me,” he says.
In the immediate aftermath of the procedure, he noticed an improvement in his symptoms: “Every day after that for about two weeks, the pain was less and less.”
Jim’s recovery time after mild® was brief. By 2 weeks post-procedure, Jim says, “I was walking. I was standing. I was pain-free.”
Today, Jim reports that he’s “back to normal,” feeling fine, and can walk and stand with “no problems at all.”
Jim encourages his fellow LSS patients to “absolutely […] not be afraid to have it done.”
“If you ever want to be able to tie your shoes again,” he says, “I’d suggest the mild® Procedure.”
Interested in speaking to a doctor who performs the mild® Procedure for spinal stenosis? Check out our mild® physician finder to locate a healthcare provider near you!
Hi, my name is Jim and I had the mild® Procedure done by Dr. Aboumerhi.
Standing was minimal, walking, I could hardly walk anywhere. Across the room was tough because you couldn’t lift your leg. It was hard to even put your leg down onto the ground. So it was tough, so that was all minimal. And the mild® Procedure was perfect for me. I mean, we were done in half hour, 45 minutes. And I would say about two weeks later, like Dr. Aboumerhi told me, I was fine, I was walking, I was standing, and I’ll say pain free.
Absolutely do not be afraid of having it done. It’s absolutely like the name says—mild, simple. Go in and out. It was good. It was done. And I’m feeling fine.
Listen to More Patient Stories Find a mild® Doctor
Disclaimer – Patient stories reflect the results experienced by individuals who have undergone the mild® Procedure. Patients are not compensated for their testimonial.
The mild® Procedure is intended to treat lumbar spinal stenosis (LSS) caused by ligamentum flavum hypertrophy. Although patients may experience relief from the procedure, individual results may vary. Individuals may have symptoms persist or evolve or other conditions that require ongoing medication or additional treatments.
Please consult with your doctor to determine if this procedure is right for you.
This case study explores the successful treatment of a patient, age 87, by Dr. Youssef Josephson. After he decided to move this patient to the mild® Procedure for lumbar spinal stenosis (LSS), Dr. Josephson found the procedure not only helped her avoid major surgery but also improve quality of life, regain functional ability, and discontinue use of opioids.
An 87-year-old female patient presented with the following complaints during her consultation:
Although she had undergone multiple interventions, her desired level of function remained unaddressed.
Dr. Josephson observed that despite her age, she exhibited considerable vitality and potential for functional improvement.
The multiple and unsuccessful interventions included physical therapy, epidural injections, and extensive dependency on medication.
She consulted with a spine surgeon, who proposed a significant open surgery involving fusion. After discussing this route with her physician, she opted to pass on open surgery due to concerns regarding recovery time and extensive anesthesia requirements.
Despite her overall good health, she preferred minimal levels of general anesthesia.
An MRI revealed buckling at the L4-L5 level along with ligamentum flavum hypertrophy.
Based on this imaging, the patient was diagnosed with spinal stenosis accompanied by intermittent neurogenic claudication, attributed to various risk factors and underlying health concerns.
Dr. Josephson determined the most appropriate treatment for her condition and desired outcome was mild®, with a focus on treating levels L4-L5.
Images submitted by Dr. Josephson
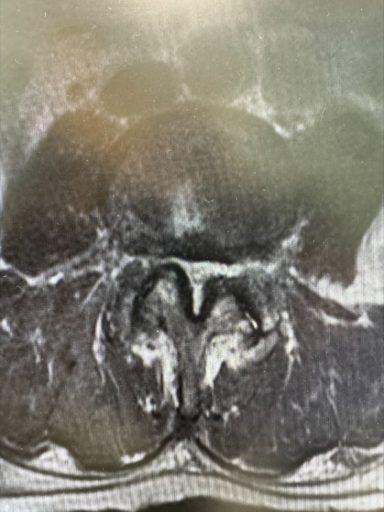
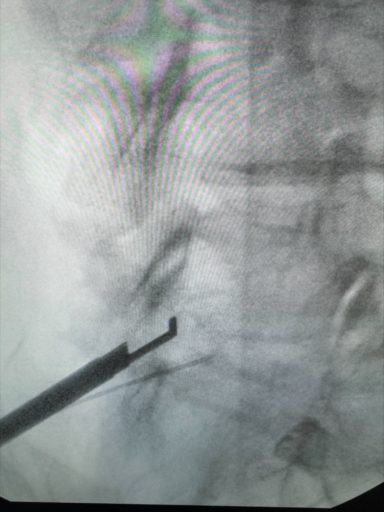
The procedure was performed utilizing fluoroscopic guidance. The patient was treated at the L4-L5 segment to address the hypertrophic ligamentum flavum which was compressing the space in the spinal canal and putting pressure on the nerves in the lower back. A mild® Tissue Sculpter was utilized to extract excess ligamentum flavum. Once satisfactory decompression was achieved, small amounts of dye and cortisone were injected to visualize improved flow and overall results.
The case concluded with a small incision closure using glue and one Steri-strip, which minimized recovery time.
At the one-week postoperative follow-up, the patient reported feeling significantly better.
At the one-month follow-up, the patient handed over her pill bag, indicating she no longer needed the medication. This is notable given that, at 87 years old, she had been reliant on opioid medication.
Dr. Josephson described this moment as the “Holy Grail” of pain management, being able to help his patient regain their quality of life and functional abilities without medication.
Dr. Josephson continues to treat spinal stenosis with mild® because the procedure provides outcomes that address both pain relief and structural issues without resorting to major surgery, interventions, or lengthy recovery periods.
As a minimally invasive procedure, mild® requires a short recovery time as well as a very minimal postoperative course. Usually within a matter of days, patients are back to themselves—indeed, a better version of themselves.
Make the move to mild® and evolve your practice’s standard of care. Learn more about how mild® can be a valuable part of your practice.
There’s good news for patients suffering from chronic lower back pain caused by lumbar spinal stenosis (LSS), as effective treatment options have become more widely available. Traditionally, administering a series of epidural steroid injections (ESIs) had been considered the standard of care, but the short-term results—typically lasting less than 6 months—left many doctors and patients looking for a more durable solution. Rather than enduring one injection after another, up to 2-3 injections per year, the minimally invasive mild® Procedure has become an increasingly sought after alternative for patients with LSS.
A recent study published in Future Medicine, highlighted some of the top reasons doctors and patients are making the move to the mild® Procedure, or, minimally invasive lumbar decompression. The mild® Procedure is a treatment option that addresses a major root cause of LSS, requires only local anesthetic and light sedation, and leaves no implant behind.
Here’s an overview about ESIs, the mild® Procedure, and how to find an interventional pain provider capable of telling you more about the procedure and its potential benefits.
Epidural steroid injections—medication injected in the lower spine to reduce swelling and offer pain relief—are typically offered to lumbar spinal stenosis patients when non-medical care methods like exercise and physical therapy have failed to provide relief.
The steroid medication in the injection is believed to reduce inflammation, which relieves pain. However, injections only treat the symptoms of stenosis and do not address the problem’s root cause.
The effects of an epidural steroid injection typically last less than 6 months so patients generally require 2-3 injections per year. Repeat steroid use is known to increase risk of infections and may cause bone loss (osteoporosis).

If epidural steroid injections aren’t effective for you, it’s important to know that your interventional pain provider may offer other treatment options. Proactively discussing and considering different options can help you and your doctor make the right treatment decision at the appropriate time.
The recent study highlighted several advantages of the mild® Procedure, including:

Finding a doctor that offers the mild® Procedure is easy. The fastest way is to use the Find a mild® Doctor feature available on this website. The finder can help you locate a mild® provider in your preferred radius of your address, city, or ZIP code.
When discussing treatment options with your doctor, be sure to ask specific questions, including:
Hear Linda’s story on how mild® relieved her pain and enhanced her everyday life.
The best time to start searching for a mild® Doctor is today. Chronic lower back pain caused by lumbar spinal stenosis typically worsens in severity over time, making early intervention crucial to restoring healthy function, movement, and quality of life. The study discussed here supports early use of the mild® Procedure, finding that:

If you’re ready to learn more about the mild® Procedure, talk to your interventional pain provider or Find a mild® Doctor today.
This case study highlights a female patient, age 67, with debilitating back pain that prevented her from completing tasks of daily living and decreased her quality of life. Despite multiple interventions, including epidural steroid injections (ESIs) and medication therapy, the patient continued to experience chronic pain. Dr. Puri, a leading pain medicine specialist at Relievus Pain Management, decided to make the move to mild® and help this patient find the lasting relief she was looking for.
The patient’s history showed degenerative disc disease and symptomatic lumbar spinal stenosis. The patient had anxiety and depression. Treatment history included ESIs, facet injections, and various medication therapy including chronic opioid therapy. The patient had no previous surgical history and was not a surgical candidate.
The patient experienced back and buttock pain and was unable to walk or stand long enough to complete daily tasks. She remained in forward flexion to relieve neurogenic claudication symptoms.
The patient experienced limited mobility and severe pain (9-10/10 pain score).
The patient set goals of:
An MRI was performed, which found multiple levels of stenosis and several spinal comorbidities.
L3-4 and L4-L5
Dr. Puri treated 2 levels using the standard approach with MAC sedation.
After the mild® Procedure, the patient was able to walk and stand twice as long. Her pain score went from 9-10/10 to 3/10 and she is now able to complete activities of daily life, including getting in and out of the shower and doing laundry without assistance.
The patient feels much happier and shares that her days are “much better.”
Listen to Dr. Puri discuss this case study.
“The patient’s mood has improved; she doesn’t use her cane, and walks up and down the stairs with greater ease. She is overall more comfortable.”
“The biggest takeaway from this case is to move to mild® sooner instead of performing repeat epidural steroid injections. This patient was already in my practice and was being overlooked. Now that we can offer mild®, we need to establish an algorithm so patients can consistently be screened and offered an opportunity to achieve the relief and functional improvement they deserve.”
Make the move to mild® and evolve your practice’s standard of care. Learn more about how mild® can be a valuable part of your practice.
Benyamin RM, Staats PS, MiDAS ENCORE Investigators. mild® is an effective treatment for lumbar spinal stenosis with neurogenic claudication: MiDAS ENCORE Randomized Controlled Trial. Pain Physician. 2016;19(4):229-242.
Mekhail N, Costandi S, Abraham B, Samuel SW. Functional and patient-reported outcomes in symptomatic lumbar spinal stenosis following percutaneous decompression. Pain Pract. 2012;12(6):417-425. doi:10.1111/j.1533-2500.2012.00565.x.
2012 data from Health Market Sciences report for Vertos Medical 2013.
Data on file with Vertos Medical.
Staats PS, Chafin TB, Golvac S, et al. Long-term safety and efficacy of minimally invasive lumbar decompression procedure for the treatment of lumbar spinal stenosis with neurogenic claudication: 2-year results of MiDAS ENCORE. Reg Anesth Pain Med. 2018;43:789-794. doi:10.1097/AAP.0000000000000868.
Based on mild® Procedure data collected in all clinical studies. Major complications are defined as dural tear and blood loss requiring transfusion.
MiDAS ENCORE responder data. On file with Vertos Medical.
Jain S, Deer TR, Sayed D, et al. Minimally invasive lumbar decompression: a review of indications, techniques, efficacy and safety. Pain Manag. 2020;10(5). https://doi.org/10.2217/pmt-2020-0037. Accessed June 1, 2020.
Deer TR, Grider JS, Pope JE, et al. The MIST Guidelines: the Lumbar Spinal Stenosis Consensus Group guidelines for minimally invasive spine treatment. Pain Pract. 2019;19(3)250-274. doi:10.1111/papr.12744.
Hansson T, Suzuki N, Hebelka H, Gaulitz A. The narrowing of the lumbar spinal canal during loaded MRI: the effects of the disc and ligamentum flavum. Eur Spine J. 2009;18(5):679-686. doi:10.1007/s00586-009-0919-7.
Treatment options shown are commonly offered once conservative therapies (e.g., physical therapy, pain medications, chiropractic) are not providing adequate relief. This is not intended to be a complete list of all treatments available. Doctors typically recommend treatments based on their safety profile, typically prioritizing low risk/less aggressive procedures before higher risk/more aggressive procedures, but will determine which treatments are appropriate for their patients.
The mild® Procedure is a minimally invasive treatment for lumbar spinal stenosis. As with most surgical procedures, serious adverse events, some of which can be fatal, can occur, including heart attack, cardiac arrest (heart stops beating), stroke, and embolism (blood or fat that migrates to the lungs or heart). Other risks include infection and bleeding, spinal cord and nerve injury that can, in rare instances, cause paralysis. This procedure is not for everyone. Physicians should discuss potential risks with patients. For complete information regarding indications for use, warnings, precautions, and methods of use, please reference the devices’ Instructions for Use.
Patient stories on this website reflect the results experienced by individuals who have undergone the mild® Procedure. Patients are not compensated for their testimonial. The mild® Procedure is intended to treat lumbar spinal stenosis (LSS) caused by ligamentum flavum hypertrophy. Although patients may experience relief from the procedure, individual results may vary. Individuals may have symptoms persist or evolve or other conditions that require ongoing medication or additional treatments. Please consult with your doctor to determine if this procedure is right for you.
Reimbursement, especially coding, is dynamic and changes every year. Laws and regulations involving reimbursement are also complex and change frequently. Providers are responsible for determining medical necessity and reporting the codes that accurately describe the work that is done and the products and procedures that are furnished to patients. For this reason, Vertos Medical strongly recommends that you consult with your payers, your specialty society, or the AMA CPT regarding coding, coverage and payment.
Vertos Medical cannot guarantee coding, coverage, or payment for products or procedures. View our Billing Guide.
Vertos is an equal employment opportunity workplace committed to pursuing and hiring a diverse workforce. We strive to grow our team with highly skilled people who share our culture and values. All qualified applicants will receive consideration for employment without regard to sex, age, color, race, religion, marital status, national origin, ancestry, sexual orientation, gender identity, physical & mental disability, medical condition, genetic information, veteran status, or any other basis protected by federal, state or local law.
Hall S, Bartleson JD, Onofrio BM, Baker HL Jr, Okazaki H, O’Duffy JD. Lumbar spinal stenosis. Clinical features, diagnostic procedures, and results of surgical treatment in 68 patients. Ann Intern Med. 1985;103(2):271-275. doi:10.7326/0003-4819-103-2-271.
Kalichman L, Cole R, Kim DH, et al. Spinal stenosis prevalence & association with symptoms: The Framingham Study. Spine J. 2009;9(7):545-550. doi:10.1016/j.spinee.2009.03.005.
Fukusaki M, Kobayashi I, Hara T, Sumikawa K. Symptoms of spinal stenosis do not improve after epidural steroid injection. Clin J Pain. 1998;14(2):148-151. doi:10.1097/00002508-199806000-00010.
Mekhail N, Costandi S, Nageeb G, Ekladios C, Saied O. The durability of minimally invasive lumbar decompression procedure in patients with symptomatic lumbar spinal stenosis: Long-term follow-up [published online ahead of print, 2021 May 4]. Pain Pract. 2021;10.1111/papr.13020. doi:10.1111/papr.13020
Friedly JL, Comstock BA, Turner JA, et al. Long-Term Effects of Repeated Injections of Local Anesthetic With or Without Corticosteroid for Lumbar Spinal Stenosis: A Randomized Trial. Arch Phys Med Rehabil. 2017;98(8):1499-1507.e2. doi:10.1016/j.apmr.2017.02.029
Pope J, Deer TR, Falowski SM. A retrospective, single-center, quantitative analysis of adverse events in patients undergoing spinal stenosis with neurogenic claudication using a novel percutaneous direct lumbar decompression strategy. J Pain Res. 2021;14:1909-1913. doi: 10.2147/JPR.S304997
Pryzbylkowski P, Bux A, Chandwani K, et al. Minimally invasive direct decompression for lumbar spinal stenosis: impact of multiple prior epidural steroid injections [published online ahead of print, 2021 Aug 4]. Pain Manag. 2021;10.2217/pmt-2021-0056. doi:10.2217/pmt-2021-0056
Abstract presented at: American Society of Pain and Neuroscience Annual Conference; July 22-25, 2021; Miami Beach, FL.
Mobility Matters: Low Back Pain in America, Harris Poll Survey, 2022. View data and full summary here.
Deer TR, Grider JS, Pope JE, et al. Best Practices for Minimally Invasive Lumbar Spinal Stenosis Treatment 2.0 (MIST): Consensus Guidance from the American Society of Pain and Neuroscience (ASPN). J Pain Res. 2022;15:1325-1354. Published 2022 May 5. doi:10.2147/JPR.S355285.